Context
Microbiome profiling via Next Generation Sequencing (NGS) techniques is rapidly changing the landscape of microbiology. However, to ensure accurate microbiome measurements, it is essential to ensure the microbiome workflow is unbiased and robust.1 For instance, it has been reported that bias in microbiomics analyses can be introduced at every step2-4 including the DNA extraction step if microbial cells are not uniformly and completely lysed. Mechanical cell lysis using beads of various composition (bead beating) is a generally accepted method used to lyse various microbes5 and, although a variety of mechanical lysis devices and instruments are available, their performance greatly varies.6-8 Until recently, there has not been a convenient way to assess the efficiency of lysis afforded by any particular instrument, however, with the release of the ZymoBIOMICS™ Microbial Community Standard (Zymo Research Corp., Irvine, CA) this is now possible. The ZymoBIOMICS™ Microbial Community is a mock microbial community standard comprised of ten well-characterized organisms (i.e., 5 gram-positive, 3 gram-negative, and 2 yeast) that are mixed at defined proportions. The standard contains microbes of different cell wall recalcitrance and of different cell size making it ideal for evaluating cell lysis efficiency. In this study, the efficiency of microbial lysis with the ZymoBIOMICS™ Microbial Community Standard was assessed using the Precellys® Evolution tissue homogenizer combined with Cryolys® Evolution cooling unit (Bertin Instruments).
Materials
- Instrument: Precellys® Evolution and Cryolys® Evolution (Bertin Instruments)
- Sample: DNA of the twelve aliquots (75 µl each) of the ZymoBIOMICS™ Microbial Community Standard extracted using the ZymoBIOMICS™ DNA Miniprep Kit (Zymo Research Corp.) following the manufacturer’s protocol.
- Buffer: 725 µl of Lysis Solution per sample included in the ZymoBIOMICS™ DNA Miniprep Kit (Zymo Research Corp.)
Protocol
Initial Control cell lysis
- The control cell lysis was performed as described in the cell lysis section using a Supplier X tissue homogenizer set at maximum speed (6.5m/s).
- Lysis of the control was optimized by using the following run:
– 5*60 sec, 5 minutes break after each minute (total time 25 min)
- DNA extraction and sequencing were performed as explained in the Cell lysis protocol section.
Cell lysis
- Cell lysis was performed using a Precellys® Evolution set at maximum speed (10,000 rpm) fitted with a Cryolys® Evolution cooling unit set at 4 °C.
- Lysis of the standard was assessed in triplicate and several run where tested in order to validate the optimal run:
– 1*60 sec (total time 1 min)
– 3*60 sec, 60 sec break after each minute (total time 5 min)
– 5*60 sec, 60 sec break after each minute (total time 9 min)
- Extracted DNA was quantified using a Qubit™ dsDNA BR Assay Kit (Thermo Fisher Scientific) and the microbial composition of the extracted DNA determined via 16S rRNA gene sequencing using an Illumina® MiSeq™ (2x300bp).
- Sequencing and data analysis were performed by the ZymoBIOMICS™ Sequencing Service (Zymo Research).
Results
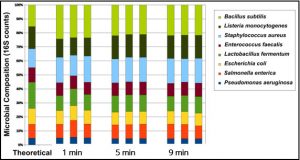
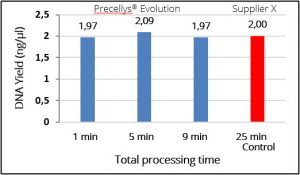
DNA quantification indicated the yields of extracted DNA reached a maximum (compared to control) when lysed for 1 minute using the Precellys® Evolution tissue homogenizer (Figure 1). This indicates that efficient cell lysation can be achieved by homogenizing cells for 1 minute instead of 5 minutes which was previously reported. These data were supported by 16S sequencing data that showed the microbial composition reflected the theoretical closely indicating that complete lysis was achieved in as little as 1 minute of bead beating (Figure 2).
In addition, standard composition is consistent and stays stable with the theoretical value even after 5 minutes of homogenization using Precellys® Evolution tissue homogenizer.
Figure 1. DNA concentrations achieved after homogenizing cells for 1, 3 and 5 minutes using the Precellys® Evolution tissue homogenizer representing a total processing time of 1, 5 and 9 minutes respectively. High yield DNA was obtained in as little as 1 minute cell lysis. The “Control” sample is the DNA yield obtained using an alternative homogenizer (Supplier X) and was achieved in approximately 5 minutes bead beating time and 25 minutes processing time in total.
Figure 2. Microbial composition analysis (16S) closely matches the ZymoBIOMICS™ Microbial Community Standard’s theoretical composition in as little as 1 minute using the Precellys® Evolution tissue homogenizer. DNA was extracted from ZymoBIOMICS™ Microbial Community Standard using the ZymoBIOMICS™ DNA Miniprep Kit and Precellys® Evolution tissue homogenizer and yields determined at the processing times indicated.
Customer
Zymo Research is a privately-held American manufacturer of molecular biology research tools used for DNA and RNA research and analysis. Their offices are headquartered at
17062 Murphy Ave., Irvine, CA 92614, Tel: 949-679-1190.
www.zymoresearch.com